Fesoterodine Fumarate ER
Toviaz
Strengths & Sizes
Note: product and packaging images are not actual size
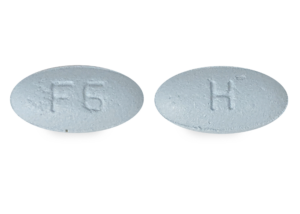
- NDC#31722-033-30
- UPC Code331722033305
- ImprintF6/H
Note: product and packaging images are not actual size
- NDC#31722-034-30
- UPC Code331722034302
- ImprintF7/H